Ongoing Projects
A Revolutionary Brain Machine Interface to Restore Functions in Amputees

Our lab have invented a novel implantable technology to sense and decode human nerve signals for restoring the lost motor functions in amputees. We are a key contributor to the DARPA Hand Proprioception and Touch Interfaces program, by innovating an ultra-low-noise chip that can suppress neural interface noise to extract signals on individual fascicles and fibers, and a deep recurrent adversarial autoencoder to decode the motor intention. Our research enables a new approach to harvest rich neural information within the nerves that leads to the world-first 15 degrees of freedom motor decoding on transradial amputee patients. We are working on a chronic clinical study, where patients learn to control a dexterous prosthetic hand in the real world as well as in virtual environments.
Patients' feedback:
“The feeling of moving a finger just like I can move the fingers on my good hand is so cool….
no it’s way more than cool. I really think you can give me a new hand.”
“This is nothing like a grasp pattern…with training for myself and working with this I can get
back to natural hand.”
“It feels real natural … it’s very close to a natural intention.”
“I am so happy to be taking part in this, I can make a difference for all the others that don’t have
the chance to be a part of this project.”
Collaborators:
Edward Keefer (physiology) from Nerves Incorporated
Jonathan Cheng (surgery) from the University of Texas Southwestern Medical Center
Catherine Zhao (machine learning) from the University of Minnesota.
Adaptive Deep Brain Stimulation to Treat Parkinson's Disease and Other Neurological Disorders
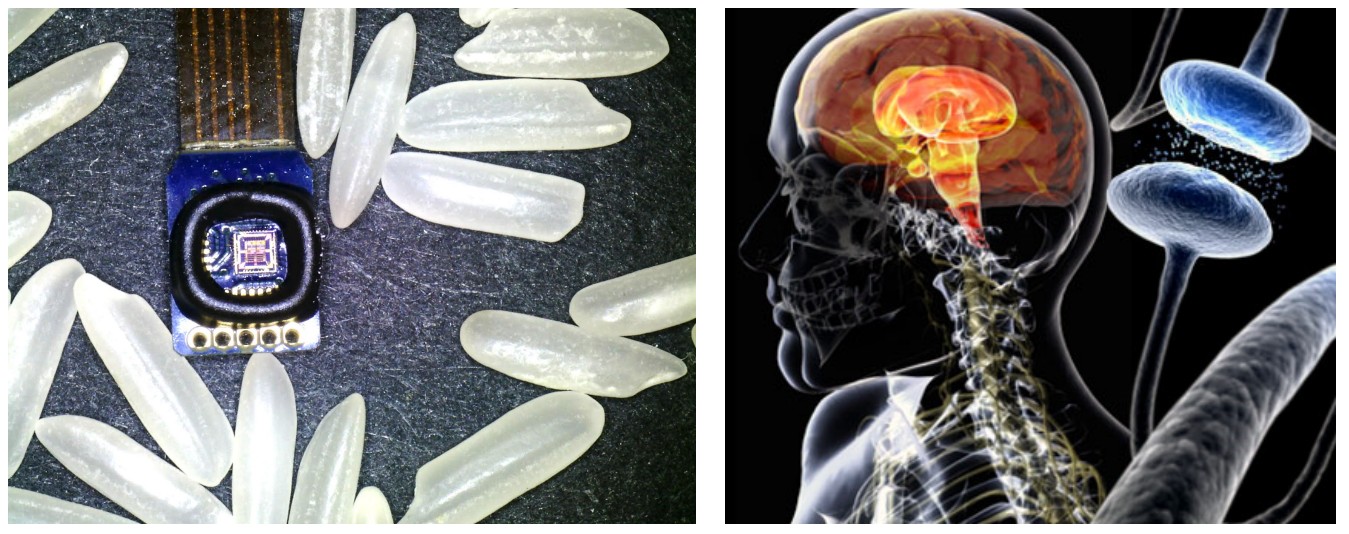
Electrical stimulation has been widely used in scientific and clinical investigations. For example, deep brain stimulation alone can treat essential tremor and Parkinson's disease, dystonia, obsessive-compulsive disorder, chronic pain, major depression, post-traumatic stress disorder, and focal epilepsy. However, there is little understanding of how electrical stimulation works and how to optimize the stimulation protocols for better treatment outcomes.
Our lab has designed a new bidirectional neural interface that can uniquely reject stimulation noise artifacts and acquire neural feedback during stimulation. Thus it becomes possible to conduct scientific experiments for learning how electrical stimulation modulates individual neurons and the neural circuits. It also allows incorporating neural feedback for improving stimulation efficacy and implementing new adaptive neuromodulation therapies. We are setting up clinical trial to test our bidirectional neural interface and evaluate the adaptive neuromodulation therapy with 5 – 10 Parkinson’s disease patients and essential tremor patients within the next two years.
Collaborators:
Hubert Lim (neurophysiology) from the University of Minnesota.
Tay Netoff (neurophysiology) from the University of Minnesota.
Michael Park (neural surgery) from the University of Minnesota.
Matthew Johnson (neurophysiology) from the University of Minnesota.
Joshua Aman (neurology) from the University of Minnesota.
Neural Recording, Stimulation, and Signal Processing ASICs

The NeuroElectronics lab innovates Application-Specific Integrated Circuit (ASIC) chips to sense, analyze, and modulate neural signals. These ASICs are enabling a number of important and groundbreaking research in brain machine interfaces and neuromodulation. The lab collaborates with medical device and neural instrumentation companies to implement the technologies into products for clinical and scientific uses.
The following new technologies have been developed based on our ASICs: (1) bidirectional cortical neural interface to support simultaneous recording and stimulation and to inform adaptive electrical stimulation. (2) Ultra-low-noise peripheral nerve neural interface and deep neural network to sense and analyze nerve neural signals. (3) A highly miniature, high-density neural interface based on analog hyper-threading and on-chip neural signal processing. (4) A charged-balanced neural stimulator based on a novel redundant sensing theory.
Collaborators:
Nerves Incorporated
Plexon
Heraeus